uniQure (QURE) Up on Safety Data From Huntington's Disease Study
Shares of uniQure N.V. QURE were up 27% on Thursday after the company announced data on its gene therapy, AMT-130, which is being developed for treating Huntington’s disease.
The company reported safety and biomarker data from 10 patients enrolled in the lower-dose cohort of its ongoing phase I/II study on AMT-130 for the given indication.
Per the company, six of the 10 enrolled patients in the study received AMT-130 and four patients received an imitation surgical procedure. The study is being conducted in the United States. Data from the same showed that treatment with AMT-130 led to a mean reduction of 53.8% of mutant HTT (mHTT) observed in cerebral spinal fluid (“CSF”) at 12 months in the given patient population.
Also, patients treated with AMT-130 had Neurofilament Light Chain (NfL) in CSF near baseline at 12 months.
Shares of uniQure have lost 5.8% so far this year compared with the industry’s decline of 25.6%.
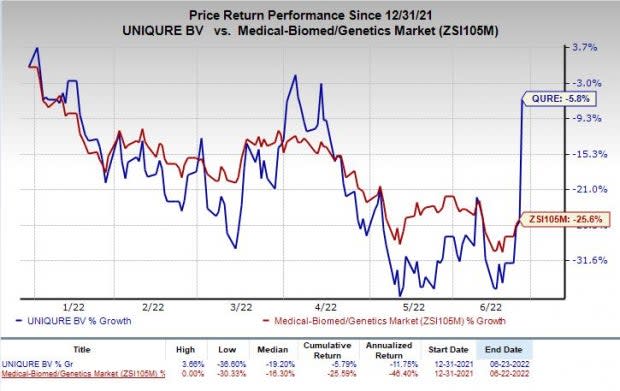
Image Source: Zacks Investment Research
The phase I/II study on AMT-130 for the treatment of Huntington’s disease is evaluating efficacy signals in 26 total patients — a 10-patient, low-dose cohort followed by a 16-patient, high-dose cohort in the United States.
uniQure plans to initiate a third cohort in the ongoing study that will explore the use of alternative stereotactic navigation systems to simplify the placement of catheters for infusions of AMT-130 later in 2022. The company plans to complete patient enrollment in the third cohort by 2023-end.
In Europe, the phase Ib/II study evaluating AMT-130 will enroll 15 patients with early manifest Huntington’s disease across two dose cohorts.
In December 2021, uniQure released initial data on the first four patients enrolled in the lower-dose cohort of its ongoing phase I/II clinical trial of AMT-130 for the treatment of Huntington’s disease.
Gene therapies are promising but complex to develop. Quite a few companies are developing gene therapies for various diseases, thus making the target market a competitive one.
Zacks Rank & Stocks to Consider
uniQure currently carries a Zacks Rank #3 (Hold). Better-ranked stocks in the biotech sector are Leap Therapeutics, Inc. LPTX, Aeglea BioTherapeutics, Inc. AGLE and Precision BioSciences, Inc. DTIL, all carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
The Zacks Consensus Estimate for Leap Therapeutics’ loss per share has narrowed 11.1% for 2022 and 5.9% for 2023 in the past 60 days.
Earnings of Leap Therapeutics have surpassed estimates in three of the trailing four quarters and missed the same on the other occasion. LPTX delivered an earnings surprise of 1.92%, on average.
Aeglea BioTherapeutics’ loss per share estimates narrowed 19.4% for 2022 and 37.5% for 2023 in the past 60 days.
Earnings of Aeglea BioTherapeutics have surpassed estimates in two of the trailing four quarters and missed the same on the other two occasions. AGLE delivered an earnings surprise of 9.47%, on average.
Precision BioSciences’ loss per share estimates narrowed 21.7% for 2022 and 31.4% for 2023 in the past 60 days.
Earnings of Precision BioSciences have surpassed estimates in each of the trailing four quarters. DTIL delivered an earnings surprise of 76.15%, on average.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 