Iovance's (IOVA) Q3 Earnings Beat Estimates, Revenues Nil
Iovance Biotherapeutics, Inc. IOVA incurred a loss of 63 cents per share in third-quarter 2022, narrower than the Zacks Consensus Estimate of a loss of 64 cents. In the year-ago quarter, the company reported a loss of 55 cents.
Without any marketed product and revenue-generating collaboration, the company did not record any revenues during the quarter.
Following the earnings announcement, shares of Iovance were up 4.8% in after-market trading on Nov 3. The stock has declined 55.3% this year compared with the industry’s 22.3% fall.
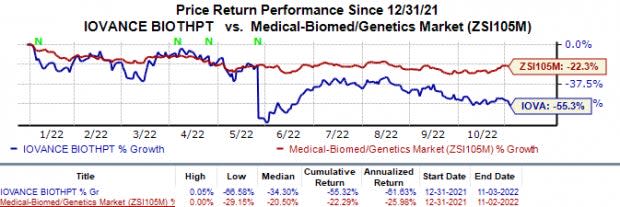
Image Source: Zacks Investment Research
Quarter in Detail
Research & development expenses were $72.5 million, up 10.9% from the year-ago quarter’s levels, primarily due to increased related personnel costs and facility building costs.
General and administrative expenses increased 33.5% from the prior-year quarter’s figure to $27.9 million due to an increase in related personnel costs and facility-related costs.
The company had $366.6 million in cash, cash equivalents, short-term investments and restricted cash as of Sep 30, 2022, compared with $430.9 million on Jun 30, 2022.
It expects the cash level to be sufficient to fund the current and planned operations in 2024.
Pipeline Updates
Iovance is developing its lead pipeline candidate, lifileucel, as a monotherapy for treating metastatic melanoma and metastatic cervical cancer in separate pivotal phase II studies — C-144-01 and C-145-04 — for metastatic melanoma and recurrent, metastatic or persistent cervical cancer, respectively, in previously-treated patients.
Following discussions with the FDA, Iovance initiated a rolling biologics license application (BLA) submission for lifileucel in August 2022, which is expected to be completed by this year’s end. If the BLA is approved, lifileucel will be the first FDA-approved individualized, one-time cell therapy for melanoma patients. It will also be Iovance’s first-ever therapy to receive FDA approval. The BLA filing will be supported by data from the pivotal Cohort 4 of the C-144-01 study, wherein patients treated with the therapy achieved an objective response rate (ORR) of 29%.
Iovance has also been engaged in discussions with the FDA for lifileucel in cervical cancer. Based on these discussions, IOVA plans to reopen enrolment in Cohort 2 of the C-145-04 study. The study will enroll patients who have previously received anti-PD-1 therapy. IOVA expects this study to support a potential BLA filing for lifileucel in cervical cancer indication.
Iovance intends to start a separate phase III study to evaluate the combination regimen of lifileucel and Merck’s MRK Keytruda (pembrolizumab) in frontline metastatic melanoma by 2022-end. The combination of lifileucel and Merck’s Keytruda, which has already been evaluated in Cohort 1A of the multi-cohort phase II study (IOV-COM-202), demonstrated an overall response rate of 67% in the given indication. This late-stage study will also serve as a confirmatory study for the pivotal Cohort 4 of the C-144-01 study.
Iovance is also evaluating another TIL therapy, LN-145, as monotherapy for head and neck squamous cell carcinoma and non-small cell lung cancer (NSCLC) in two separate studies.
The company is also evaluating LN-145 in combination with Merck’s Keytruda or Bristol Myers’ BMY Opdivo/Yervoy in different cohorts of the IOV-COM-202 study. The study is evaluating LN-145 plus Merck’s Keytruda or Bristol-Myers’ Opdivo/Yervoy in separate cohorts in NSCLC patients.
Opdivo and Yervoy are two blockbuster immunotherapies marketed by Bristol Myers.
This October, Iovance started dosing participants in the phase I/II IOV-GM1-201 study to evaluate its new TIL candidate, IOV-4001, in patients with previously treated advanced melanoma and metastatic NSCLC in two separate cohorts.
IOV-4001 leverages the TALEN technology licensed from Cellectis S.A. CLLS to genetically knock out PD-1 in TIL cells. This license is part of a collaboration finalized between Iovance and Cellectis in 2020. Per the agreement terms, IOVA has an exclusive worldwide license to use Cellectis’ proprietary TALEN technology to develop gene-editing TIL therapies targeting cancer. In return, Cellectis will receive potential milestones and royalty payments for products developed using the TALEN technology.
Iovance Biotherapeutics, Inc. Price

Iovance Biotherapeutics, Inc. price | Iovance Biotherapeutics, Inc. Quote
Zacks Rank
Iovance currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report
Cellectis S.A. (CLLS) : Free Stock Analysis Report
Iovance Biotherapeutics, Inc. (IOVA) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 