Aeglea (AGLE) Plunges 26% After Rare Disorder Drug Study Fails
Aeglea BioTherapeutics AGLE announced data from the interim analysis of a phase I/II study of pegtarviliase for the treatment of cystathionine beta synthase deficiency, also known as Classical Homocystinuria. The stock nosedived 26.21% on Wednesday after the company reported that the study failed to achieve its objectives.
The candidate, pegtarviliase, is a novel recombinant human enzyme engineered to reduce elevated levels of total homocysteine in patients with Classical Homocystinuria. It is a rare inherited disorder of methionine metabolism that leads to elevated levels of total homocysteine.
In the past year, shares of Aeglea have plummeted 90.4% compared with the industry’s 14.2% fall.
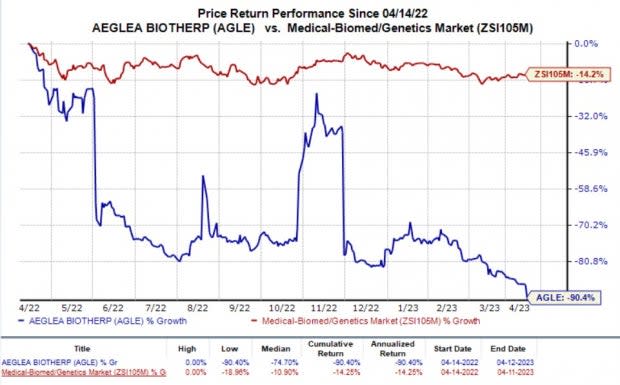
Image Source: Zacks Investment Research
The total patient population in the ongoing phase I/II dose escalation study of pegtarviliase has been divided into three cohorts. Participants in each cohort received pegtarviliase once a week for four weeks to assess safety and tolerability as well as to gather data on efficacy and optimum dose levels.
The study enrolled a total of 13 patients out of which 11 participants completed dosing and were considered evaluable. One participant withdrew from the study due to personal reasons.
Per the interim results of the study, data from the first two cohorts (0.15 mg/kg and 0.45 mg/kg, respectively) showed that treatment with pegtarviliase lowered total homocysteine levels in participants when compared with baseline values. However, in the third cohort (1.35 mg/kg), a consistent reduction in total homocysteine levels compared with baseline was not observed.
Further analysis of the data showed that anti-drug antibodies were found in the participants belonging to the third cohort. Experts speculated that this could have impacted the pharmacokinetics and reduced the effect of pegtarviliase in reducing total homocysteine levels.
All things considered, Aeglea concluded that the available data suggest exploration of higher doses or dosing of longer duration might be necessary to better determine the potential efficacy of pegtarviliase in lowering total homocysteine.
The candidate was overall well tolerated during the study, with most adverse events being mild-to-moderate injection site reactions and hypersensitivity reactions. There was one serious adverse event which was assessed as unrelated to treatment. No participants withdrew from the study or had a dose reduction due to an adverse event.
Management remains confident of the potential of its Homocystinuria treatment candidate, but unfortunately does not have compelling data to support immediate dialogue with regulatory bodies on a pivotal study design.
In the same press release, Aeglea confirmed entering the process of exploring strategic alternatives. This will include but are not limited to, an acquisition, merger, reverse merger, other business combination, sales of assets or other strategic transactions. The company further reiterates that this is not a guarantee of any transaction.
Aeglea has also not provided a date of completion of this strategic review, withholding a comment on the status of this process, for the time being. The company has slashed its workforce, including members from upper management, retaining approximately 10 employees required to support the process and continue ongoing value-creating activities.
Aeglea BioTherapeutics, Inc. Price and Consensus

Aeglea BioTherapeutics, Inc. price-consensus-chart | Aeglea BioTherapeutics, Inc. Quote
Zacks Rank and Stocks to Consider
Aeglea currently has a Zacks Rank #3 (Hold).
Some better-ranked stocks in the same industry are Aptinyx APTX, Acadia Pharmaceuticals ACAD and ADMA Biologics ADMA, all carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
In the past 90 days, the Zacks Consensus Estimate for Aptinyx’s 2023 loss per share has narrowed from 77 cents to 47 cents. In the past year, shares of Aptinyx have fallen by 88.1%.
APTX beat estimates in each of the trailing four quarters, delivering an average earnings surprise of 6.56%.
In the past 90 days, the Zacks Consensus Estimate for Acadia Pharmaceuticals’ 2023 loss per share has narrowed from 87 cents to 56 cents. In the past year, shares of Acadia Pharmaceuticals have fallen by 21.9%.
ACAD beat estimates in two of the trailing four quarters, missing the mark on other two occasions, delivering an average negative earnings surprise of 6.33%.
In the past 90 days, the consensus estimate for ADMA Biologics’ 2023 loss per share has narrowed from 19 cents to 14 cents. In the past year, shares of ADMA Biologics have increased by 55.2%.
ADMA beat estimates in three out of the trailing four quarters, delivering an average earnings surprise of 2.88%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
ACADIA Pharmaceuticals Inc. (ACAD) : Free Stock Analysis Report
Aeglea BioTherapeutics, Inc. (AGLE) : Free Stock Analysis Report
Aptinyx Inc. (APTX) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 