| Previous close | N/A |
| Open | N/A |
| Bid | N/A |
| Ask | N/A |
| Strike | N/A |
| Expiry date | N/A |
| Day's range | N/A - N/A |
| Contract range | N/A |
| Volume | |
| Open interest | N/A |
 GuruFocus.com
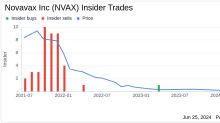
GuruFocus.comInsider Sale: Director James Young Sells Shares of Novavax Inc (NVAX)
The transaction was documented in a recent SEC Filing. Following this sale, the insider now owns 61,760 shares of Novavax Inc. Novavax Inc is a biotechnology company focused on the discovery, development, and commercialization of innovative vaccines to prevent serious infectious diseases.
 Zacks
ZacksNovavax (NVAX) Seeks Nod for Updated COVID-19 Jab in Europe
Novavax (NVAX) seeks marketing authorization in the European Union for an updated version of its COVID-19 vaccine, which has been formulated to target the JN.1 variant.
 PR Newswire
PR NewswireNovavax Submits Application to European Medicines Agency for Updated Protein-based 2024-2025 Formula COVID-19 Vaccine
Novavax, Inc. (Nasdaq: NVAX), a global company advancing protein-based vaccines with its Matrix-M™ adjuvant, today announced that it has filed for a type II variation of existing Marketing Authorization with the European Medicines Agency (EMA) for its JN.1 COVID-19 vaccine (NVX-CoV2705) for individuals aged 12 and older. The submission is in line with guidance from EMA and the World Health Organization to target the JN.1 lineage this fall.1,2