Imugene Limited (IMU.XA)
| Previous close | 0.0830 |
| Open | 0.0830 |
| Bid | 0.0000 x N/A |
| Ask | 0.0000 x N/A |
| Day's range | 0.0780 - 0.0850 |
| 52-week range | 0.0390 - 0.1900 |
| Volume | |
| Avg. volume | 5,679,533 |
| Market cap | N/A |
| Beta (5Y monthly) | 3.21 |
| PE ratio (TTM) | N/A |
| EPS (TTM) | -0.0050 |
| Earnings date | N/A |
| Forward dividend & yield | N/A (N/A) |
| Ex-dividend date | N/A |
| 1y target est | N/A |
 GlobeNewswire
GlobeNewswireBile Tract Cancer expansion study opens following clearance of Imugene’s MAST trial high dose cohort
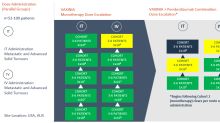
The expansion trial is expected to enrol 10 patients with bile tract cancers (cholangiocarcinoma)Interim results from the MAST trial have demonstrated positive responses in gastrointestinal cancers, particularly in cholangiocarcinoma where one patient treated with CF33-hNIS (VAXINIA) achieved a complete response and another patient achieved stable diseaseThe fifth cohort in the intratumoural (IT) arm of the VAXINIA monotherapy trial has now been cleared with no safety signals seen to date; the c
 GlobeNewswire
GlobeNewswireImugene's oncolytic virotherapy VAXINIA and B cell immunotherapy HER-Vaxx featured at the AACR Annual Meeting 2024
Oncolytic virus CF33-hNIS (VAXINIA) alone or in combination with KEYTRUDA is a safe treatment option for advanced cancer patients.Encouraging VAXINIA efficacy, including a complete response (CR) and two partial responses (PRs), has warranted cohort expansions in biliary tract cancer and other tumour types.Compared to chemotherapy alone, vaccination with HER-Vaxx was associated with a 40% overall survival benefit.HER-Vaxx induced HER2-specific antibody levels correlate with tumour reduction. SYDN
 GlobeNewswire
GlobeNewswirePhase 1 onCARlytics solid tumour trial advances to combination arm treatment
The OASIS trial is a first-in-class clinical trial combining a CD19-expressing oncolytic virus, which flags solid tumours for attack by CD19-targeting therapeutics such as Amgen’s Blincyto®, Gilead’s Yescarta® and Imugene’s own allogeneic CAR T Azer-celOASIS potentially represents a paradigm shift in solid tumour treatmentCohort Review Committee (CRC) observed no safety issues in the onCARlytics monotherapy lead-in study and recommended opening of the combination arm of the studyOvarian, breast