Sarepta (SRPT) Down on Clinical Hold on DMD Candidate SRP-5051
Sarepta Therapeutics SRPT announced that the FDA has placed a clinical hold on its next-generation exon-skipping pipeline candidate, SRP-5051 (vesleteplirsen), following a report of a serious adverse event of hypomagnesemia in the ongoing phase II MOMENTUM study. The study is evaluating SRP-5051 for treating Duchenne muscular dystrophy (DMD) patients with skipping exon 51.
The FDA has requested information on all cases of hypomagnesemia, including a small number of non-serious grade 2 cases, following treatment with SRP-5051. The regulatory authority has requested these adverse events data to assess the adequacy of the risk mitigation and safety monitoring plan. Sarepta is planning to submit these data to the FDA and resume dosing in the study as soon as possible. Please note that hypomagnesemia can lead to life-threatening complications.
Sarepta claimed that the occurrence of hypomagnesemia was transient and the magnesium levels returned to normal in patients following additional supplementation.
Shares of Sarepta declined 3.9% during after-hours trading on Jun 23, following the dismal regulatory update related to SRP-5051. In fact, Sarepta’s shares have declined 20.3% so far this year compared with the industry’s decrease of 25.6%.
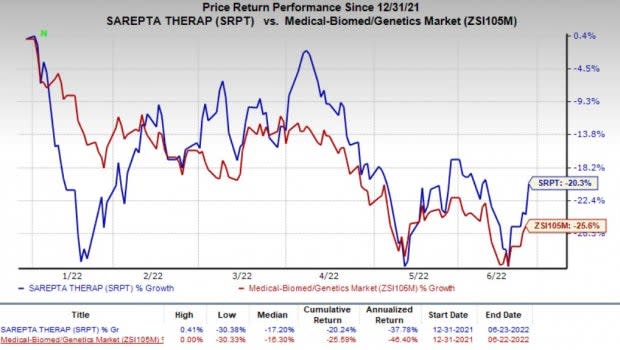
Image Source: Zacks Investment Research
SRP-5051 is being evaluated in a similar patient population targeted by Sarepta’s lead drug Exondys 51. SRP-5051 has the potential to offer DMD patients a better alternative with a much lower dose and a new drug with longer patent protection.
SRP-5051 achieved a 1.6-fold increase in exon skipping and a five-fold increase in the percentage of normal dystrophin compared to Exondys 51 at 24 weeks, per previous data readout from the MOMENTUM study. Moreover, the PROMOVI study evaluated a weekly 30mg/kg of Exondys 51, significantly higher compared to the SRP-5051 dose.
The clinical hold placed by the FDA only affects the U.S. portion of the MOMENTUM study, which is being conducted globally. The study continues in ex-U.S. regions where ongoing enrollment is expected to be completed by the end of 2022.
Sarepta has a portfolio of three commercialized DMD drugs — Exondys 51, Vyondys 53 and Amondys 45. The strong performance of these DMD drugs has been the key driver for Sarepta in the past few years.
Sarepta also puts focus on developing novel treatments for DMD as well as other muscular atrophies. Apart from the next-generation SRP-5051, the company is developing several gene therapies targeting muscular atrophies. Sarepta is also one of the pioneers in gene therapy development.
Last year, SRPT initiated the first pivotal study — EMBARK — to evaluate its lead gene therapy candidate, SRP-9001, for DMD patients. Sarepta is developing the micro-dystrophin-encoding gene therapy candidate in collaboration with Roche RHHBY. Sarepta and Roche entered into a licensing agreement to develop SRP-9001 in 2019. Per the agreement, Roche has exclusive rights to launch and commercialize SRP-9001 in the ex-U.S. markets. Roche made $1.15-billion upfront payments and will pay up to $1.7 billion in regulatory and sales milestones to Sarepta.
Apart from DMD, Sarepta is developing gene therapies for treating Limb-girdle muscular dystrophy, Mucopolysaccharidosis type IIIA (MPS IIIA) and Pompe Disease in different clinical-stage studies.
Sarepta Therapeutics, Inc. Price
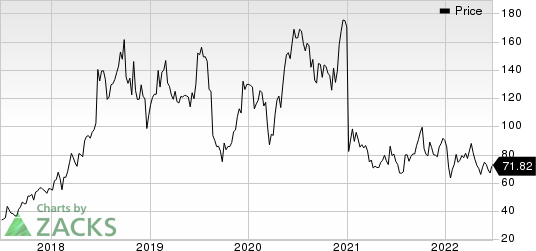
Sarepta Therapeutics, Inc. price | Sarepta Therapeutics, Inc. Quote
Zacks Rank & Stocks to Consider
Sarepta currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
A couple of better-ranked biotech stocks are Alkermes ALKS and Sesen Bio SESN, both sporting a Zacks Rank #1.
The Zacks Consensus Estimate for Alkermes’ 2022 loss per share has narrowed from 10 cents to 3 cents in the past 60 days. Shares of ALKS have risen 27.1% year to date.
Earnings of Alkermes beat estimates in each of the last four quarters, the average being 350.48%.
The Zacks Consensus Estimate for Sesen Bio’s 2022 loss has narrowed from 33 cents to 32 cents per share in the past 60 days. Shares of SESN have gained 1.2% in the year-to-date period.
Earnings of Sesen Bio beat estimates in three of the last four quarters and missed the mark on one occasion, the average surprise being 69.94%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 