Regulus (RGLS) Up as FDA Accepts IND for Kidney Disease Drug
Regulus Therapeutics Inc. RGLS announced that the FDA has accepted the investigational new drug (“IND”) application for its novel pipeline candidate, RGLS8429, seeking approval to begin clinical studies on the same for the treatment of autosomal dominant polycystic kidney disease (“ADPKD”).
The company plans to initiate a phase I study evaluating RGLS8529 in healthy volunteers for treating ADPKD later in the second quarter of 2022.
Shares of Regulus were up 54.8% following the announcement of the news on Wednesday. The stock has lost 15.6% so far this year compared with the industry’s decline of 26.8%.
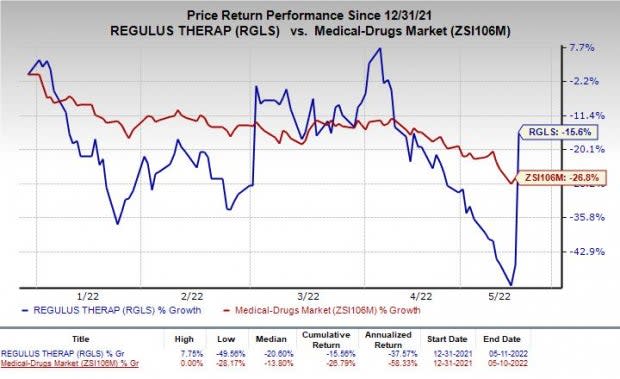
Image Source: Zacks Investment Research
A phase I single-ascending dose study will assess the safety, tolerability and pharmacokinetics of RGLS8429 in healthy volunteers. After this study, Regulus intends to begin a phase Ib multiple-ascending dose study in adult patients with ADPKD.
The company plans to report data from the healthy volunteer study and start dosing patients with ADPKD during the second half of 2022.
Per the press release, in the United States, around 140,000 individuals are diagnosed with ADPKD, with an estimated four to seven million affected worldwide. Hence if successfully developed and upon potential approval, RGLS8429 may offer a transformative treatment option for the given patient population.
Apart from RGLS8429, Regulus is developing another pipeline candidate, lademirsen (RG-012), for the treatment of adult patients with Alport syndrome, an inherited form of kidney disease.
The company, under its collaboration with French pharma giant, Sanofi SNY, announced the completion of enrollment in the phase II HERA study evaluating lademirsen for treating Alport syndrome in February 2022.
In November 2018, Regulus transitioned development responsibilities for RG-012 to Genzyme, a subsidiary of Sanofi. SNY bears all costs for the development of lademirsen.
RGLS currently has no approved product in its portfolio. Therefore, the successful development of its pipeline candidates remains in key focus for the company.
Zacks Rank & Other Stocks to Consider
Regulus currently carries a Zacks Rank #2 (Buy). Other stocks worth considering in the same sector include Onconova Therapeutics, Inc. ONTX and Soleno Therapeutics, Inc. SLNO, both carrying the same Zacks Rank #2 at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Onconova Therapeutics’ loss per share estimates have narrowed 22.4% for 2022 and 18.2% for 2023 over the past 60 days.
Earnings of Onconova Therapeutics have surpassed estimates in each of the trailing three quarters. ONTX delivered an earnings surprise of 22.72% on average.
Soleno Therapeutics’ loss per share estimates have narrowed 9.1% for 2022 and 25.9% for 2023 over the past 60 days.
Earnings of Soleno Therapeutics have surpassed estimates in two of the trailing four quarters, met the same once and missed the same on the other occasion. SLNO delivered an earnings surprise of -10.92%, on average.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Sanofi (SNY) : Free Stock Analysis Report
Regulus Therapeutics Inc. (RGLS) : Free Stock Analysis Report
Onconova Therapeutics, Inc. (ONTX) : Free Stock Analysis Report
Soleno Therapeutics, Inc. (SLNO) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 