Pharma Stock Roundup: ABBV to Buy CERE, JNJ's 2024 View, LLY & NVS Get FDA Nod
This week, AbbVie ABBV announced a definitive agreement to acquire neuroscience drugmaker Cerevel Therapeutics for $8.7 billion. The FDA approved Novartis’ NVS rare blood disorder drug, Fabhalta (iptacopan) and Eli Lilly’s LLY Jaypirca for its second leukemia indication. J&J JNJ announced sales and earnings growth expectations for 2024 and the 2025-2030 period. Pfizer PFE decided not to move forward with phase III studies on its twice-daily weight loss pill, danuglipron, due to side effects seen in a phase IIb study.
Recap of the Week’s Most Important Stories
AbbVie to Buy Cerevel Therapeutics: AbbVie offered to buy Cerevel Therapeutics for $45 per share in cash, which adds up to a total equity value of approximately $8.7 billion. The acquisition will strengthen AbbVie’s neuroscience pipeline by adding CERE’s novel pipeline candidatesbeing studied across a range of psychiatric and neurological disorders, including schizophrenia, Parkinson's disease (PD) and mood disorders.
Cerevel’s key pipeline candidates are emraclidine (phase II for schizophrenia), tavapadon (phase II for PD) and darigabat (phase II for epilepsy and panic disorder). Data from studies on emraclidine, darigabat and tavapadon are expected in 2024. The transaction is expected to close in mid-2024, subject to shareholder and regulatory approval. The boards of directors of both companies have approved the transaction.
Pfizer’s Obesity Drug Not to Move to Phase III Following Side-Effects: Pfizer announced top-line data from a phase IIb study evaluating its twice-daily oral GLP-1R agonist candidate, danuglipron, in adults with obesity. Top-line data from the study showed that twice-daily dosing of danuglipron resulted in mean placebo-adjusted weight reductions ranging from -8% to -13% at 32 weeks and -5% to -9.5% at 26 weeks. However, high rates of gastrointestinal side effects were observed in the study.
Discontinuation rates were greater than 50%, seen across all doses, much higher than approximately 40% with placebo. Pfizer thus decided not to move forward with phase III studies on the twice-daily formulation of danuglipron. The pharmacokinetic study of the once-daily formulation of danuglipron continues. Depending on the outcome of the once-daily formulation, it will decide on the path forward.
The failure of the phase IIb study on twice-daily oral formulation of danuglipron is a significant blow to Pfizer’s efforts to take a share of the growing market of GLP-1 drugs for treating obesity.
FDA Approves Novartis’ Fabhalta (iptacopan) for PNH: The FDA approved Novartis’ iptacopan for the treatment of adults with paroxysmal nocturnal hemoglobinuria (PNH), a rare and chronic blood disorder. Iptacopan, to be marketed by the name of Fabhalta, became the first oral monotherapy approved for treating adults with PNH. Fabhalta’s approval is based on data from the APPLY-PNH study in adults with PNH and anemia despite prior anti-C5 treatment and supported by the APPOINT-PNH study in complement inhibitor-naïve patients.
Novartis expects to launch Fabhalta for PNH in December.The drug is also being evaluated for a range of complement-mediated diseases, including immunoglobulin A nephropathy (IgA nephropathy), C3 glomerulopathy (C3G), immune complex membranoproliferative glomerulonephritis (IC-MPGN) and atypical hemolytic uremic syndrome (aHUS).
FDA Approves Lilly’s Jaypirca for 2nd Indication: The FDA granted accelerated approval to Lilly’s BTK inhibitor Jaypirca (pirtobrutinib) for the second indication, chronic lymphocytic leukemia (CLL). Jaypirca/pirtobrutinib was approved for the first indication, mantle cell lymphoma, in the United States in January 2023. The approval of Jaypirca (100 mg & 50 mg tablets)is for adult patients with CLL or small lymphocytic lymphoma who have received at least two lines of therapy, including a BTK inhibitor and a BCL-2 inhibitor. The decision was based on data from the phase I/II BRUIN study, which showed that treatment with Jaypirca led to an overall response rate of 72% in the above-mentioned patient group.
J&J’s 2024 and Long-Term Outlook: J&J announced that it expects operational sales growth in the range of 5-6% in 2024. Adjusted earnings are expected in the band of $10.55-$10.75 per share in 2024, which represents operational growth of 7.3% at the mid-point.
In 2025, operational sales are expected to grow around 3% due to the expected launch of biosimilar versions of its blockbuster immunology drug, Stelara, in the year. Stelara recorded sales of more than $8 billion in the first nine months of 2023.
J&J expects sales to pick up thereafter at a compound annual growth rate (CAGR) of 5-7% for the 2025-2030 period. In its Innovative Medicine unit, J&J expects operational sales to witness a CAGR of 5-7% for the 2025-2030 period. In the MedTech segment, operational sales are expected to grow in the upper range of its market, which is projected to rise 5-7% through 2027.
The FDA granted Breakthrough Therapy Designation (BTD) to J&J’s pipeline candidate, TAR-200, for treating patients with Bacillus Calmette-Guérin (BCG)-unresponsive, high-risk non-muscle-invasive bladder cancer (HR-NMIBC) who are ineligible for, or decline, radical cystectomy. The BTD is based on data from the phase IIb SunRISe-1 phase IIb study. Data from the study presented at the European Society for Medical Oncology showed that treatment with TAR-200 led to a 77% complete response rate in the above-mentioned patient group.
Roche to Acquire Obesity Drugmaker: Roche entered into a definitive agreement to acquire private biotech, Carmot Therapeutics, which is making subcutaneous and oral incretins to treat obesity. Carmot Therapeutics’ lead pipeline candidate is CT-388, which is a dual GLP-1/GIP receptor agonist being developed for treating obesity in patients with and without type II diabetes. Data from a phase Ib study on lead pipeline candidate, CT-388, demonstrated substantial weight loss, which suggests it can treat obesity with a differentiated profile.
It is also developing a once-daily oral, small molecule GLP-1 receptor agonist (CT-996 in phase I) and a once-daily subcutaneous injectable, dual GLP-1/GIP receptor agonist (CT-868 in phase II) for obesity.
For the acquisition, Roche will make a payment of $2.7 billion to Carmot Therapeutics in cash. In addition, Carmot Therapeutics will be entitled to potential milestone payments of up to $400 million.
The NYSE ARCA Pharmaceutical Index declined 0.59% in the last five trading sessions.
Large Cap Pharmaceuticals Industry 5YR % Return
Large Cap Pharmaceuticals Industry 5YR % Return
Here’s how the eight major stocks performed in the last five trading sessions.
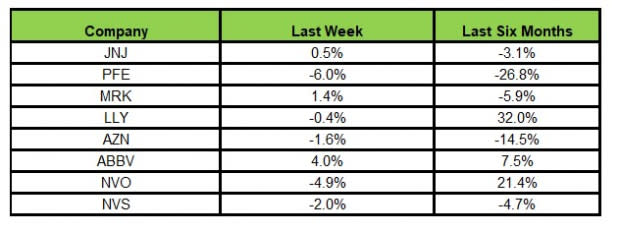
Image Source: Zacks Investment Research
In the last five trading sessions, AbbVie rose the most (4.0%) while Pfizer declined the most (6.0%).
In the past six months, Lilly has risen the most (32.0%), while Pfizer has declined the most (26.8%).
(See the last pharma stock roundup here:ABBV to Buy Immunogen, NVS Ups Mid-Term Sales View & More)
What's Next in the Pharma World?
Watch for pipeline and regulatory updates next week.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Johnson & Johnson (JNJ) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
AbbVie Inc. (ABBV) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 